Mesenchymal stem cells (MSCs) are among the most researched cells for regenerative therapies due to their diversified capabilities and promising outcomes in various conditions. This article outlines the three most common sources of MSCs and their primary mechanisms of action.
The most common sources of MSCs include:
- Bone Marrow: A rich source of MSCs, widely used in clinical applications.
- Adipose Tissue: Easily accessible and abundant, offering a viable alternative to bone marrow.
- Umbilical Cord Tissue: A non-invasive source with potent regenerative properties.
MSCs work primarily through:
- Differentiation: The ability to develop into specific cell types needed for tissue repair.
- Immunomodulation: Modulating the immune response to reduce inflammation and promote healing.
- Paracrine Signaling: Releasing growth factors and cytokines that aid in tissue regeneration and repair.
Understanding these sources and mechanisms help highlight the therapeutic potential of MSCs in regenerative medicine.
What are mesenchymal stem cells?
Stem cells are the body’s raw materials — cells from which all other cells with specialized functions are created. Mesenchymal stem cells are adult stem cells that have self-renewal, immunomodulatory, anti-inflammatory, signaling, and differentiation properties.
Mesenchymal stem cells (MSCs) self-renewal capacity is characterized by their ability to divide and develop into multiple specialized cell types present in a specific tissue or organ.
Mesenchymal stem cells (MSCs) can be sourced from a variety of tissue including adipose tissue (fat), bone marrow, umbilical cord tissue, blood, liver, dental pulp, and skin.
Clinical Applications
MSCs are widely used in the treatment of various diseases due to their self-renewability, differentiation, anti-inflammatory, and immunomodulatory properties. In-vitro (performed in a laboratory setting) and In-vivo (taking place in a living organism) studies have supported the understanding mechanisms, safety, and efficacy of MSC therapy in clinical applications. 1
According to a 2022 study conducted by Molnar et al. 1
“MSCs mainly exert their regenerative effects through paracrine and endocrine modes of action, which include immunomodulatory, anti-inflammatory, mitogenic, anti-apoptotic, anti-oxidative stress, anti-fibrotic, and angiogenic influences.” 1
Adipose Tissue-Derived MSCs (ADSCs)
Adipose tissue-derived MSCs are obtained from the subcutaneous adipose tissue (fat tissue), they can be rapidly acquired in large numbers and with high cellular activity through a liposuction procedure. 2
ADSCs are likely to be more viable when sourced from a younger donor. This may prove to be an issue with older patients that participate in an autologous procedure (using your cells), as the older cells may be less fitted for long-term survival in the recipient. Adipose tissue-derived MSCs (ADSCs) from younger donors present a higher proliferation rate (survivability after transplant) when compared to elderly ones, but the differentiation capacity is maintained with aging, thus having advantages on bone marrow mesenchymal stem cells (BM-MSCs).
ADSCs maintain their potential to differentiate into cells of mesodermal (middle cell layer) origin and are well known for their low immunogenicity and immunomodulatory effects. Less than 1% of these cells express the HLA-DR protein on their surface, contributing to their immunosuppressive properties and making them suitable for clinical applications in allogeneic transplantation and the treatment of resistant immune disorders. It is widely accepted that ADSCs can be used to treat a variety of conditions, including orthopedic issues such as spinal cord injury, arthritis, localized joint inflammation, knee pain, and other musculoskeletal problems.
Despite their promise, several challenges remain regarding the clinical use of ADSCs. These include limitations in cell proliferation related to donor age, restricted differentiation potential in some cases, and the lack of standardized protocols for their isolation and application.
According to a study published by Mazini et al 2
“ADSCs represent many therapeutic challenges in terms of origin, type, and the manner to use them, different recent investigations pave the way to their successful therapeutic use in tissue repair. More insights into standardizing technical use are warranted to evaluate the in-depth efficacy and safety of ADSCs-based therapy and evaluate the benefit-to-risk ratio in clinical applications. The beneficial effects of stem cells, and there with the paradigm of tissue regeneration, may not be restricted to cellular restoration, but may also be related to the transient paracrine actions of the cells.”
Bone Marrow-Derived MSCs (BM-MSCs)
Bone marrow-derived mesenchymal stem cells (BM-MSCs), which are classified as multipotent adult stem cells, are widely used in the treatment of various diseases via their self-renewable, differentiation, and immunomodulatory properties.
“In-vitro and in-vivo studies have supported the understanding mechanisms, safety, and efficacy of BM-MSCs therapy in clinical applications. The number of clinical trials in phase I/II is accelerating; however, they are limited in the size of subjects, regulations, and standards for the preparation and transportation, and administration of BMSCs, leading to inconsistency in the input and outcome of the therapy.” 3
Limitations
Bone marrow harvesting is indeed an invasive and potentially painful procedure that typically requires anesthesia. BM-MSCs, or bone marrow-derived mesenchymal stem cells, are a rare population, representing only about 0.002% of the total stromal cell population. Their isolation depends on factors such as the patient’s condition and the volume of bone marrow collected. 2
Like ADSCs, bone marrow stem cell’s quantity and quality decline with age. BM-MSCs are likely to be more viable when sourced from a younger donor when employing an allogeneic (cells come from a third party) treatment. This may prove to be an issue with older patients that participate in an autologous procedure, as the older cells may be less fitted for long-term survival in the recipient. This issue is outlined by Chu et al. in a 2020 study. 3
“Stem cells that were isolated from elders had a low rate of proliferation and differentiation ability into osteoblasts, whereas they increase the expression of apoptosis markers and SA-β-gal positive cells (an indicator of the senescence cells)” 3
Most of the preclinical and clinical trials have shown promising results of BMSCs on the treatments of various diseases with few adverse effects during follow-up periods. Currently, BM-MSCs therapy has been used in the treatment of osteoarthritis, neurodegenerative diseases, and sports-related injuries.
Umbilical Cord Tissue-Derived Mesenchymal Stem Cells (UC-MSCs)
UC-MSCs can be sourced from a variety of areas including Wharton’s Jelly, cord lining, and peri-vascular region of the umbilical cord. As a commonly discarded tissue, the umbilical cord contains a rich source of mesenchymal stromal cells, which are therefore obtained non-invasively. 4
“UC-MSCs are the most primitive type of MSCs, shown by their higher expression of Oct4, Nanog, Sox2, and KLF4 markers.” 4
— Torres Crigna et al 2018
Umbilical cord tissue-derived mesenchymal stem cells have the ability to differentiate into different cell types and have the greatest proliferation rate of the three mentioned types of stem cells (adipose, bone marrow, cord tissue).
Similar to adipose tissue and bone marrow-derived MSCs, UC-MSCs are known to secrete growth factors, cytokines, and chemokines, improving different cell repair mechanisms. These functions all assist the anti-inflammatory and immunomodulatory properties of MSCs.
Non-invasive cell products
The harvesting procedure of UC-MSCs is non-invasive as it does not require extraction from the patient. The MSCs are taken directly from an area of an ethically donated human umbilical cord.
UC-MSCs also have a high proliferative potential than BMSCs and ASCs meaning they expand in vitro more effectively allowing for greater efficiency when obtaining higher cell numbers.5
Studies have found that UC-MSCs genes related to cell proliferation (EGF), PI3K-NFkB signaling pathway (TEK), and neurogenesis (RTN1, NPPB, and NRP2) were upregulated (increase in the number of receptors) in UC-MSCs compared to in BM-MSCs.
| Characteristics | BMSCs | ADSCs | UC-MSCs |
| Harvesting procedure | Invasive | Invasive | Non-invasive |
| Effect of age on cell quantity and quality | Declines with donor age | Declines with donor age | Unaffected |
| Potency to differentiate into nerve cells | Lower | Lower | Higher |
| Cell renewal capabilities | Lower proliferative potential | Lower proliferative potential | Higher proliferative potential |
| Expression of embryonic markers | Lower | Lower | Higher |
| Immune modulatory properties | Good | Good | Good |
| Allogenetic cell rejection | No | No | No |
| Ethical issues | No | No | No |
| Risk of tumorigenicity (promoting tumor growth) | No | No | No |
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7510077/
How do mesenchymal stem cells work in the body?
Mesenchymal stem cells utilize their self-renewal, immunomodulatory, anti-inflammatory, signaling, and differentiation properties to influence positive change within the body. Mesenchymal stem cells (MSCs) also have the capacity to self-renew by dividing and developing into multiple specialized cell types present in a specific tissue or organ. Mesenchymal stem cells are adult stem cells, meaning they present no ethical concerns, MSCs are not sourced from embryonic material.
“The characteristics of presenting no major ethical concerns, having low immunogenicity, and possessing immune modulation functions make MSCs promising candidates for stem cell therapies.”
Jiang, et al.6

Ready to get started?
Fill out a brief patient screening application and find out if you are a candidate for our stem cells therapies.
Immunomodulatory (regulating the immune system)
Mesenchymal stem cells (MSCs) can regulate the immune system by promoting an inflammatory response when the immune system is under-activated and reducing inflammation when the immune system is overactivated. MSCs can play a key role in preventing the immune system from attacking itself similar to what one may see in many autoimmune disorders. According to a 2013 study conducted by Bernardo et al. MSCs, when exposed to sufficient levels of pro-inflammatory markers (cytokines) respond by promoting an immune-suppressive response to dampen inflammation and promote tissue homeostasis. 6
According to a 2019 study conducted by Jiang, et al.
This balance is outlined by the same study in the figure below.
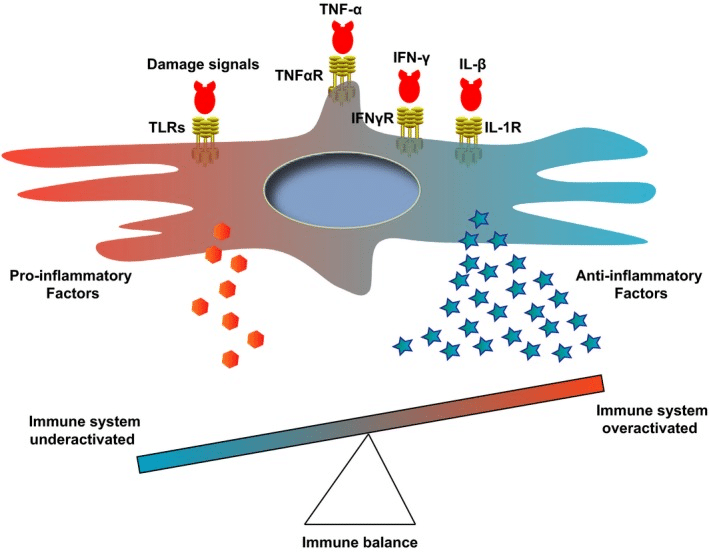
Anti-inflammatory (reducing harmful inflammation)
Inflammation is a response from the immune system that is aimed at protecting the body from harmful external stimuli as well as aid and repair the body. However, when dysregulated inflammation can have a detrimental effect on the body. An immune system that is dysregulated for an extended period can lead to a variety of autoimmune conditions such as Multiple Sclerosis, Type 1 Diabetes, Inflammatory Bowel Disease, or Lupus.
The anti-inflammatory properties of MSCs play a key role in their therapeutic abilities. 7
¿How MSCs reduce inflammation?
MSCscan be administered through various routes—including local, intravenous, or intra-lymphatic injections—taking advantage of their homing properties to target inflammation and tissue injury. Both autologous and allogeneic MSCs have demonstrated safety and efficacy in numerous animal models, with no significant immune rejection or hypersensitivity even after repeated administrations. Their immunomodulatory capacity has been extensively documented: in lung injury models, MSCs decreased the protein levels of pro-inflammatory cytokines including IL-1β, IL-6, and TNF-α, reduced neutrophil recruitment, and improved histological outcomes. In myocarditis, liver injury, renal inflammation, wound healing, GVHD, and IBD models, MSCs consistently reduced inflammatory markers, improved tissue regeneration, and enhanced survival outcomes. These effects are often mediated by shifts in cytokine expression and immune cell recruitment, such as promoting IL-4 and reducing IL-2 and INF-γ. Additionally, the health status of the recipient influences MSC immunomodulatory function—for instance, decreasing Th1 responses in GVHD and SLE, while shifting Th2 to Th1 responses in allergic diseases. This highlights the importance of tailoring MSC therapy to the patient’s immune condition to maximize therapeutic outcomes. 8
According to a 2020 study conducted by Gugjoo et al. 8
“In general, the central role of mesenchymal stem cells (MSCs) in maintaining homeostasis (immuno-modulation and anti-inflammatory activities) occurs by interacting with immune cells and is mediated through cytokines, chemokines, cell surface molecules, and metabolic pathways. MSCs suppress T-cell proliferation, cytokine secretion, and cytotoxicity
Gugjoo et al. 8
MSC secretome and extracellular vesicles (exosome signaling)
The regenerative effects of mesenchymal stem cells do not solely rely on their differentiation potential and ability to replace the injured tissues but also mediated by their secretome via paracrine mechanisms.
MSC secretome is a set of bioactive factors that are released into the body including cytokines, growth factors, extracellular vesicles, neurotrophins, soluble proteins, lipids, and nucleic acids.
The secretomes that are released play important roles in the regulation of many physiological processes and they are of increasing interest as potential biomarkers and therapeutic targets in diseases. 9
According to a 2016 study conducted by Arutyunyan et al. UC-MSCs exhibit increased secretion of neurotrophic factors such as bFGF, nerve growth factor (NGF), neurotrophin 3 (NT3), neurotrophin 4 (NT4), and glial-derived neurotrophic factor (GDNF) compared to bone marrow-derived (BM-MSCs) and adipose tissue-derived (AT-MSCs).
Otherwise, according to Peshkova et al. 10, umbilical cord-derived MSCs’ conditioned media demonstrated the highest cytokine and growth factor levels and, despite exhibiting a mostly pro-inflammatory cytokine profile, were able to promote anti-inflammatory macrophage polarization. This shift toward the M2 macrophage phenotype plays a fundamental role in regenerative processes, as M2 macrophages are associated with tissue repair, resolution of inflammation, angiogenesis, and remodeling of the extracellular matrix—key components in achieving effective healing and regeneration.
Additionally, UC-MSCs secrete significantly higher amounts of several important cytokines and hematopoietic growth factors, including G-CSF, GM-CSF, LIF, IL-1α, IL-6, IL-8, and IL-11, compared to BM-MSCs. This suggests that UC-MSCs may be more potent than other sources of MSCs. 10
Homing properties (how MSCs know where to go)
One of the key benefits of MSC is their ability to target specific areas of concern due to their intrinsic homing capabilities. Mesenchymal stem cell homing, when administered systemically can be defined as exiting circulation and migrating to the injury site. (
According to a 2019 study conducted by Ullah et al. 11
Systemic homing is a multistep process governed by specific molecular interactions. “The process of systemic homing can be split into five steps: (1) tethering and rolling, (2) activation, (3) arrest, (4) transmigration or diapedesis, and (5) migration”.
This process is outlined in the figure below.
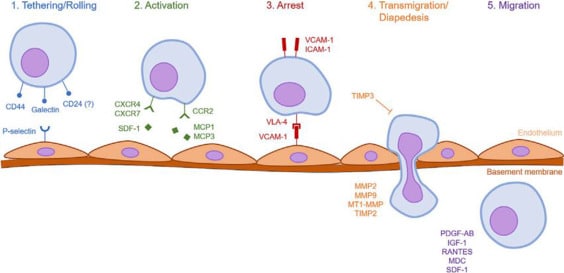
Differentiation (becoming new types of cells)
Mesenchymal stem cells are multipotent stem cells that can self-renew and differentiate into different cell types. In other words, mesenchymal stem cells can become a variety of different cell types including; adipose tissue, cartilage, muscle, tendon/ligament, bone, neurons, and hepatocytes. 12
According to a 2016 study conducted by Almalki et al. – “The differentiation of MSCs into specific mature cell types is controlled by various cytokines, growth factors, extracellular matrix molecules, and transcription factors (TFs). 12
Mesenchymal stem cells contribute to tissue regeneration and differentiation, including the maintenance of homeostasis and function, adaptation to altered metabolic or environmental requirements, and the repair of damaged tissue. 13
Conclusion
There is a growing body of scientific evidence supporting the remarkable therapeutic potential of mesenchymal stem cells (MSCs). Numerous studies have demonstrated their ability to self-renew, modulate the immune system, reduce inflammation, communicate with surrounding cells, and differentiate into various tissue types. These properties allow MSCs to play a pivotal role in the treatment of a wide range of degenerative, inflammatory, and immune-related conditions.
Among the different MSC sources, umbilical cord tissue-derived MSCs (UC-MSCs) are emerging as a superior option due to their youthful profile, higher proliferation capacity, low immunogenicity, and potent biological activity. Recent research suggests that UC-MSCs may offer greater therapeutic efficacy compared to MSCs from adult tissues, making them a promising choice for next-generation regenerative therapies.
At Iuvenis Clinic of Panama, we offer advanced regenerative treatments using expanded UC-MSCs, sourced from ethically donated umbilical cord tissue, processed in a GMP-compliant laboratory approved by Panama’s Ministry of Health (MINSA).
We invite you to explore the full range of therapies we offer by visiting our Treatments section. If you’re ready to take the next step, you can also find out if you are a candidate for stem cell therapy here.
Discover how regenerative medicine with UC-MSCs could help improve your quality of life—safely, ethically, and effectively—with Iuvenis Clinic.
References
- Molnar et al. (2022). Mesenchymal Stem Cell Mechanisms of Action and Clinical Effects in Osteoarthritis: A Narrative Review. PubMed ↩︎
- Mazini et al. (2019). Regenerative Capacity of Adipose-Derived Stem Cells (ADSCs), Comparison with Mesenchymal Stem Cells (MSCs). PMC ↩︎
- Chu et al (2020) An Update on the Progress of Isolation, Culture, Storage, and Clinical Application of Human Bone Marrow Mesenchymal Stem/Stromal Cells. PubMed ↩︎
- Torres Crigna et al. (2018). Stem/stromal cells for treatment of kidney injuries with focus on preclinical models. PMC ↩︎
- Arutyunyan et al. (2016). Umbilical Cord as Prospective Source for Mesenchymal Stem Cell-Based Therapy. PubMed ↩︎
- Jiang et al (2020,) Immune modulation by mesenchymal stem cells. Cell proliferation. PubMed ↩︎
- Ryu et al (2020) Application of Mesenchymal Stem Cells in Inflammatory and Fibrotic Diseases. International journal of molecular sciences. PubMed ↩︎
- Gugjoo et al (2020). Mesenchymal Stem Cell-Mediated Immuno-Modulatory and Anti- Inflammatory Mechanisms in Immune and Allergic Disorders. PubMed ↩︎
- Nazarie Ignat et al. (2021). Regenerative Potential of Mesenchymal Stem Cells’ (MSCs) Secretome for Liver Fibrosis Therapies. PubMed ↩︎
- Peshkova et al. (2023) MSCs’ conditioned media cytokine and growth factor profiles and their impact on macrophage polarization. PubMed ↩︎
- Ullah et al. (2019). Mesenchymal Stromal Cell Homing: Mechanisms and Strategies for Improvement. PubMed ↩︎
- Almalki, S. G., & Agrawal, D. K. (2016). Key transcription factors in the differentiation of mesenchymal stem cells. Differentiation; research in biological diversity. PubMed ↩︎
- Grafe Et al. (2018) TGF-β Family Signaling in Mesenchymal Differentiation. PubMed ↩︎